In practice, chemists simplify the notation by using a bracketed noble gas symbol to represent the configuration of the noble gas from the preceding row because all the orbitals in a noble gas are filled. 
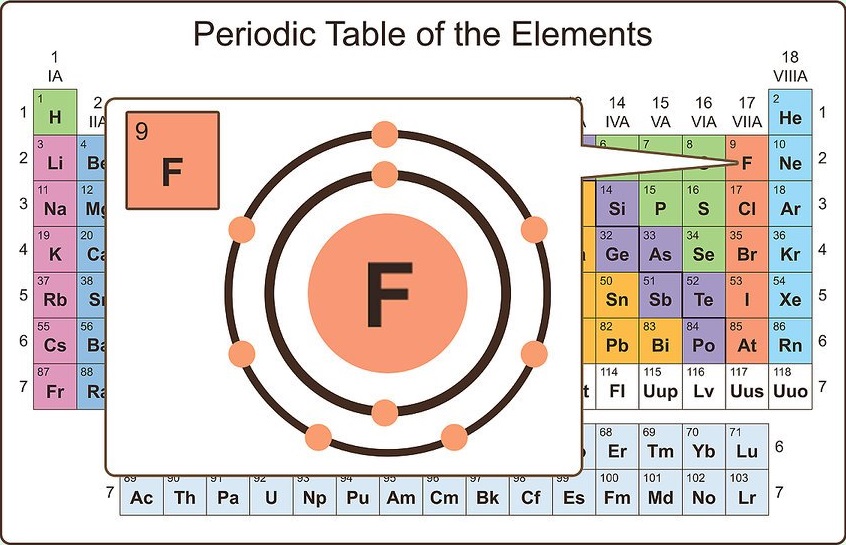
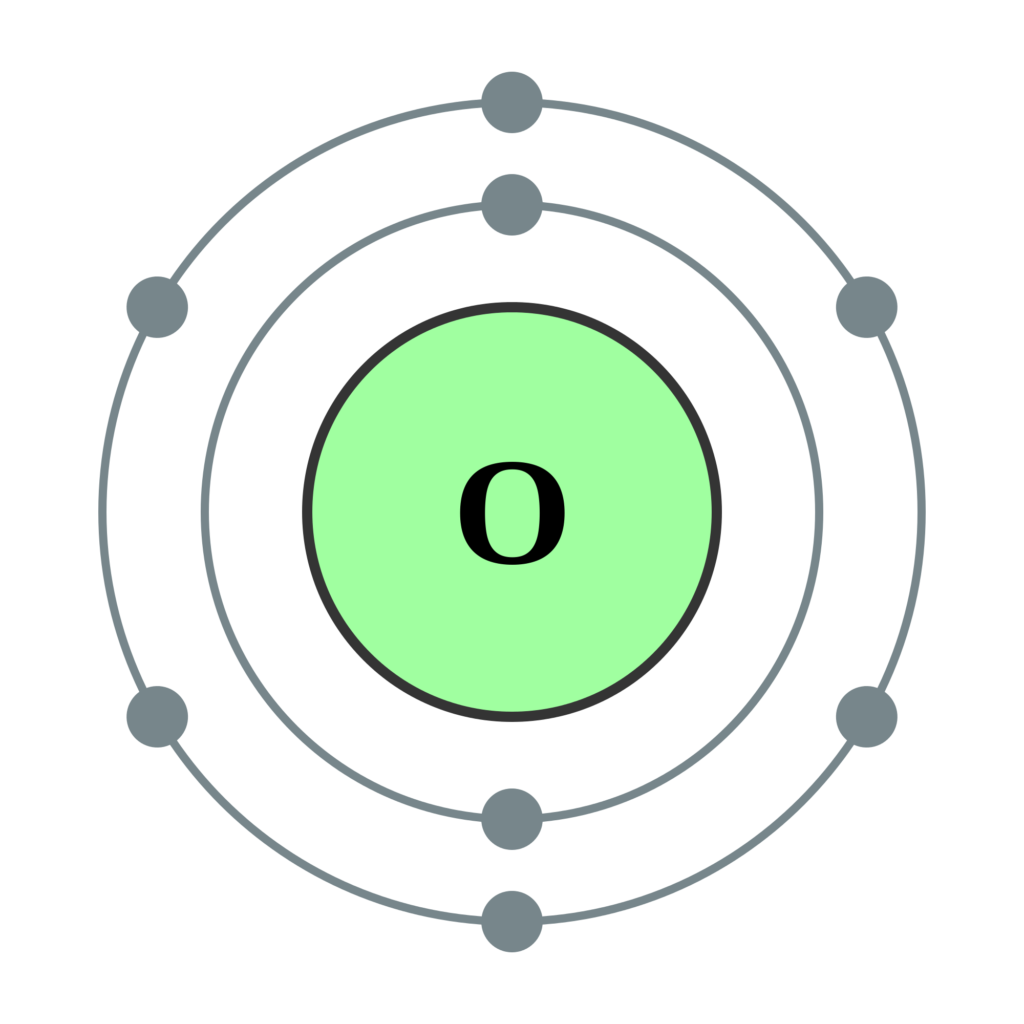
When we reach neon, with Z = 10, we have filled the 2 p subshell, giving a 1 s 22 s 22 p 6 electron configuration:Īs we continue through the periodic table in this way, writing the electron configurations of larger and larger atoms, it becomes tedious to keep copying the configurations of the filled inner subshells. Similarly, fluorine has the electron configuration 1 s 22 s 22 p 5: Fluorine has one electron pair in the 1 s orbital, one electron pair in the 2 s orbital, and 2 electrons pairs with one unpaired electron in the 2 p orbital. Oxygen has one electron pair in the 1 s orbital, one electron pair in the 2 s pair, and one electron pair, and 2 unpaired electrons in the 2 p orbitals. Because all the 2 p orbitals are degenerate, it doesn’t matter which one has the pair of electrons. One electron must be paired with another in one of the 2 p orbitals, which gives us two unpaired electrons and a 1 s 22 s 22 p 4 electron configuration. The electron configuration of nitrogen is thus 1 s 22 s 22 p 3.Īt oxygen, with Z = 8 and eight electrons, we have no choice. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
